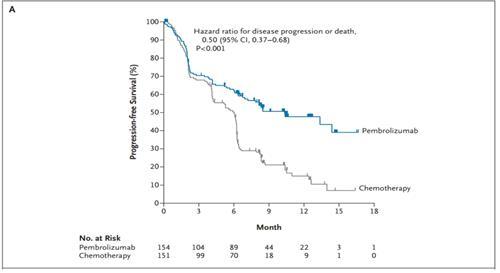
Resultsģ05 pts were randomized (pembro, 154 chemo, 151). For this analysis, response/PD was assessed by investigators per RECIST v1.1. Endpoints included PFS (primary) OS, ORR, and safety (secondary) and duration of response (exploratory). Pts randomized to pembro who completed 2 years of therapy or who stopped pembro after achieving CR and then had PD were eligible for a second course of pembro monotherapy. Pts randomized to chemo who had PD and met eligibility criteria could cross over to pembro monotherapy. 
Randomization was stratified by ECOG PS (0/1), histology (squamous/nonsquamous), and region (East Asia/other). MethodsĮligible pts were randomized to pembro (200 mg Q3W for up to 35 cycles ) or chemo. We report updated efficacy and safety from KEYNOTE-024 with 5 years follow-up. Superiority of 1L pembro monotherapy vs chemo in pts with metastatic NSCLC with PD-L1 TPS ≥50% and no sensitizing EGFR/ALK alterations was demonstrated in KEYNOTE-024 (NCT02142738).
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
